I recently offered 25,000 dollars in prizes to anyone who could disprove that Dr. Luis Sanchez committed multiple felonies based on documents that provide direct evidence of a chargeable crime. No additional evidence is needed. So far no one has been knocking at my door and nary a call from anyone.
How can we interpret this? Are the prizes not worthy of the time involved in such an endeavor? No, I think there is something for everyone including 2oo volumes of the Classics in Medicine. A current listing on Amazon is listing 187 volumes for almost $5000 and in addition to this I am offering dozens of 1970s skateboards, 2 vintage Schwinn Krate bikes, autographs from polio vaccine pioneers Sabin and Salk, multiple signed and one-of-a-kind items related to Boston politician James Michael Curley, a menu, cocktail menu, matchbook and other very rare items from Boston’s Cocoanut Grove which was the site of the deadliest nightclub fire in history in 1942, signed lithographs and prints from Warhol and Keith Haring and a signed poster from Adam Ant. So the prizes are not chopped liver and burnt toast. The prizes are worthy so in a time or work/benefit analysis the benefits are high. What about the time or work involved? Would the work and time it takes to disprove this claim make it not worth the effort like trying to prove some logic or mathematical theorem? The James Randi Educational Foundation offered 1 million dollars to anyone who could prove that homeopathy works but no one has claimed it yet. Why? Because homeopathy does not work. My challenge is fairly simple as the documents are a quick read and the claims direct and specific. All one has to do is read the documents in search of acts that might be considered crimes and use a legal reference to see if they correspond to criminal code. If your examination of the documents finds everything is on the up-and-up or just one or two acts commensurate with felonies then the prizes are yours. A simple task that should take less than an hour or two. So why is no one knocking at my door? No one is knocking for the same reason the million dollar homeopathy reward hasn’t been claimed. The reason no one is knocking at my door is because Luis Sanchez did in fact commit multiple felonies. I could double the prizes, give you everything I own and offer my very soul but there would still be no one knocking.
So the question for us is this, how is it possible that an individual contracted by a state agency could commit felony crimes that are documented by direct evidence and not be called on it? The crimes include flagrant forensic fraud no different from Annie Dookhan who faked, forged and fabricated some 60,00 samples at the Massachusetts crime lab. Fabricating evidence is fabricating evidence so how is it possible that this fabrication of evidence has not been identified and addressed by anyone? First of all there is no direct oversight or independent oversight to state physician health programs (PHPs). Not one agency exists that can accept a complaint, investigate it and punish offenders. Second, there is an inability or unwillingness to investigate or punish the offenders for various reasons. PHPs have portrayed themselves as allies to law enforcement and have requested they defer to them any issues related to physicians. When crimes are reported to local and state police and other law-enforcement agencies they are deemed the illusory product of a diseased mind. States Attorneys General legally represent occupation board. Policy dictates they side with medical boards so individual doctors are rebuffed if they ask for help. The FBI will not investigate. I was told unless the AGO requested it or a public outcry ensued they would not address any issues regarding physician health programs including laboratory fraud and diagnosis rigging. This inability or unwillingness to meaningfully investigate the offenders is a pervasive problem and represent a total systems failure. No one is minding the minders and no one wants to mind the minders. The agencies that could and should investigate crimes are taking a “not-my-problem” stance. The medical boards have agreed to absolute deference to PHPs and additionally have individuals or groups within the board who are able to block, deflect and otherwise dismiss information implicating the state PHP. All of the documents seen below were provided to Physician Health and Compliance Board counsel Deb Stoller beginning in 20111 but she apparently kept all the documents to herself. Her ethical obligation when being provided with evidence of forensic fraud and other crimes was to notify the proper authorities. She did not. She suppressed all of the information with direct evidence of crimes, an act that is much more grave and vile than Dookhan as the documents show collusion between the MA PHP and one of the drug testing labs used by almost all state PHPs. The documents provide direct evidence of lab fraud and falsification of evidence that is clear and specific. No alternative explanations exist. Concealing this type of misconduct just allows it to continue and what is seen here is not rare. I have heard from many many doctors who claim they were given bogus tests at this very same lab. Sadly I have heard of many suicides related to these very same tests. Faced with the “not my problem” stance of agencies that could and should do something but did not these doctors became hopeless and helpless and killed themselves out of despair. Their locus of control was completely lost and their was no lifeline. Selective application of the law is the root of government corruption and destroys the moral authority of the government. It inevitably leads to collapse and chaos. The Massachusetts Board of Registration in Medicine has ignored these documents for five years and assert that the PHP has not committed any crimes because they have not been charged with any crimes. Nonsense and logical fallacy. The PHP did commit crimes and so too did Board attorney Deb Stoller when she was given direct evidence of them–misprision of a felony in multiples.
Ignoring misconduct not only allows it to continue it allows it to spread. The plan is to spread these same non-FDA approved of unknown validity and easily manipulated with no accountability to other populations including students and kids. It was also recently suggested that the organization Luis Sanchez is past president of, the Federation of State Physician Health Programs (FSPHP) be put in charge of national public policy for not just doctors but for everyone.
The simple fact is that doctors are being subject to diagnostic rigging and forensic fraud and nobody cares. This is by design and fueled by the anti-doctor sentiment spread by these same groups with propaganda meant to spread the bogus dangers of a hidden cadre of drug addled and besotted doctors. It doesn’t exist but the stature of doctors in the public eye has suffered because of the mythology they created. Don’t get me wrong as any doctor with a substance abuse problem or addiction should be removed from practice and not be able to practice until safe to do so. But that is not what is happening here.
The majority of doctors being monitored by state physician health programs do not have a problem. They have loosened the diagnostic criteria so that essentially anyone referred to them is given a diagnosis and treated. Why? Because it is a money maker. All costs are out-of-pocket and no insurance is involved. The “PHP-approved” assessment centers all charge around $ 4500 for an “assessment” which inevitably leads to a 3-month inpatient stay for treatment to the tune of about $80,000. This is then followed by a five-year monitoring contract with random drug and alcohol testing 1-3 x per week. That is a total of 260 to 780 tests and the laboratory developed tests are not cheap. You do the math. The drug and alcohol assessment and testing industry makes a pretty penny off this operation and they want to bring it to you-It is in fact a Trojan horse.
So by ignoring this type of misconduct it will only get worse. People tend not to get concerned unless it impacts them. This will. Someday you or a family member may be in this same situation. It boggles my mind that doctors can be given diagnoses when they do not meet the criteria for the given diagnosis and this is confirmed by outside independent experts. This occurs because those giving the diagnosis have no accountability. They have removed themselves. But in actual fact this is political abuse of psychiatry. It is egregious.
Diagnosing someone with a disease they do not have should be a never event. The perpetrators should not only have their licenses removed but they should be punished for the crime. Forensic fraud should also be a never-event but as seen below it is done without fear of sanction-by fax no less. Giving false diagnoses and fabrication of drug and alcohol tests are actions that should never be tolerated. Not even once. How can this be happening in the United States of America? Unless people speak up this will only escalate. Below is a detailed analysis of the multiple crimes used for the $25,000 challenge. Give it a read and try to disprove the claim that Luis Sanchez committed multiple felonies. If you can you win all the prizes. If you can’t then I ask that you be outraged and do something about it as you or someone you love may be next. Ignoring this type of misconduct is not going to make anything better.
Fraud is distinguished from negligence, ignorance, and error by virtue of the fact that it is intentional. Fraudulent intent is established by examining the documentation of decisions and behaviors of those involved.
Innocent parties don’t normally alter documents or conceal information.
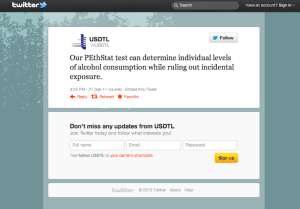
Innocent parties don’t normally “update” donor ID numbers or lab reports to “reflect that chain of custody be maintained as requested in the document below faxed from Physician Health Services, Inc. (PHS) to United States Drug Testing Laboratories (USDTL) on July 19th, 2011.
The lab report PHS is referring to is seen below which is positive for the alcohol biomarker phosphatidylethanol at a level of 365.4 ng/ml (cutoff level for a positive test is 20 ng/ml.
Note both the chain of custody and Donor ID # are listed as 461430 and there is no collection date, collectors name or location listed. The specimen was received on July 8, 2011 and reported on July 14, 2011 according to the document. On July 19th, 2011 Mary Howard requests that a collection date of July 1st, 2011 be added and that the Donor ID # be changed from 461430 to #1310.
What is the significance of this? My unique identifier for for random drug and alcohol testing is #1310 and I had this same test drawn on July 1st, 2011.

The document are from the USDTL “Litigation Packet” received December 3, 2011 which can be seen here: USDTL-LitigationPacket12:2011
USDTL “updated” the test. The document below shows they added my ID # and a collection date of July 1, 2011.
It is the responsibility of the Medical Review Officer (MRO) to make the final determination as to whether or not the positive result reported by the laboratory is reported to anyone else.
The previous Medical Director of the Massachusetts PHP, Physicians Health Services, Inc. (PHS) for fourteen years, Dr. Luis Sanchez, is also a past President of the FSPHP although PHS did not enter into formal partnership with the FSPHP until 2013. The Director of Program Operations at PHS, Linda R. Bresnahan, MS, is the current Secretary of the FSPHP.
PHS, inc. exclusive use of out of state treatment programs has been challenged. In the past PHS, Inc. has adamantly refused to allow physicians to be evaluated in State despite it being one of the medical hubs of the country. Physicians in Massachusetts who are referred for evaluation in Kansas, Georgia, Alabama, and West Virginia as PHS has convinced the Board that only these facilities are ‘experienced in the assessment and treatment of health care professionals.” In actual fact the medical directors of all of these preferred facilities are also ASAM physicians with the vast majority being in 12-step “recovery” themselves and a close colleague if not friend of the medical director of the state PHP.

“A body of men holding themselves accountable to nobody ought not to be trusted by anybody.”
― Thomas Paine
“Our minds tell us, and history confirms, that the great threat to freedom is the concentration of power. Government is necessary to preserve our freedom, it is an instrument through which we can exercise our freedom; yet by concentrating power in political hands, it is also a threat to freedom. Even though the men who wield this power initially be of good will and even though they be not corrupted by the power they exercise, the power will both attract and form men of a different stamp.”
—Milton Friedman
Dr. Luis Sanchez, M.D. served as the Medical Director of Physician Health Services, Inc. (PHS) for 15-years. Massachusetts state Physician Health Program (PHP) for 15-years. He retired in March 2013 and Dr. Steven Adelman . Sanchez also served as President of the national organization for state PHPS, the Federation of State Physician Health Programs (FSPHP).
The “PHP-blueprint” is being promoted as a “new paradigm” with a success rate twice that of other treatment programs (80%). With numbers this high, the argument goes, why bother with anything else? The attached slides are from a presentation Sanchez gave at a legal issues conference. His presents the ACGME definition of professionalism that includes competencies in altruism, accountability, excellence, duty, honor and integrity, and respect for others. Sanchez also addressed these topics in an article he wrote for the Journal of the American Medical Association (JAMA) entitled Disruptive Behaviors Among Physicians. In thiis article he discusses a “medical culture of safety” with “clear expectation and standards.” Sanchez affirms the importance of values and codes-of-conduct in the practice of medicine, and calls on physician leaders to “commit to professional behavior.”
PHS Director of Operations Linda R. Bresnahan has also been involved in leadership positions at the FSPHP including acting a Executive Secretary and Interim Executive Director.
PHPs recommend referral of physicians if there are any concerns such as getting behind on medical records. As PHS Associate DirectorJudith Eaton explains “when something so necessary is not getting done, it is prudent to explore what else might be going on.” If the PHP feels that doctor needs an assessment they will send that doctor to a “PHP-approved” facility “experienced in the assessment and treatment of health care professionals.” The physician must comply with any and all recommendations of the assessment center. To assure this the physician must sign a monitoring contract with the PHP (usually five years). USDTL is one of the labs PHPs have contracted with for forensic drug and alcohol testing.
USDTL drug testing laboratory claims to advance the”Gold Standard in Forensic Toxicology.” “Integrity: Results that you can trust, based on solid science” is listed as a corporate value. “Unlike other laboratories, our drug and alcohol testing begins and ends with strict chain of custody.” “When people’s lives are on the line, we don’t skip steps.” Joseph Jones, Vice President of Laboratory Operations explains the importance of chain-of-custody in this USDTL video presentation.
Forensic Drug and Alcohol Tests: The Need For Integrity and Accountability of the Sample
“Forensic” drug-testing differs from “clinical” drug-testing in how the results are used. “Clinical” tests are used for medical purposes in diagnosing and treating a patient.
A “forensic” test is used for non-medical purposes. It is not used for patient care, but for detecting licit and illicit substances in those who should not be using them. Pre-employment and employee assistance and professional monitoring programs are examples.
Forensic testing is held to a higher standards because the consequences of a positive result can be grave and far reaching. A positive forensic test can result in loss of rights of the individual being tested and his or her loved ones. Mistakes are unacceptable.
The Federation of State Medical Boards Policy on Physician Impairment supports this position stating “chain-of-custody forensic testing is critical” (page 14) and the “use of a Medical Review Officer (MRO) for screening samples and confirming sample results” (page 21).
Any and all drug testing requires chain-of-custody. The custody-and-control form is given the status of a legal document because it has the ability to invalidate a test that lacks complete information. Chain-of-custody provides assures specimen integrity. It provides accountability.
 The job of the MRO is to ensure that the drug testing process is followed to the letter and reviews the Custody and Control form for accuracy. The MRO also rules out any other possible explanations for a positive test (such as legitimately prescribed medications). Only then is the test reported as positive.
The job of the MRO is to ensure that the drug testing process is followed to the letter and reviews the Custody and Control form for accuracy. The MRO also rules out any other possible explanations for a positive test (such as legitimately prescribed medications). Only then is the test reported as positive.
The legal issues involved in forensic testing mandate MRO review. According to The Medical Review Officer Manual for Federal Workplace Drug Testing Programs
“the sole responsibility of the MRO is to”ensure that his or her involvement in the review and interpretation of results is consistent with the regulations and will be forensically and scientifically supportable.”
“Fatal flaws” such as lack of chain-of-custody form, missing tamper proof seal, missing signatures, or a mismatch of the sample ID and chain of custody ID invalidate the test. It is not reported. Tight chain-of-custody and MRO review is critical for the accountability and integrity of the sample.
The Medical Review Officer Certification Council provides a certification process for MROs. They also follow their own Code of Ethics. In accordance with these standards PHS has an MRO to review all positive tests. As added assurance the FSPHP guidelines state that all positive tests must be approved by the Medical Director.
also follow their own Code of Ethics. In accordance with these standards PHS has an MRO to review all positive tests. As added assurance the FSPHP guidelines state that all positive tests must be approved by the Medical Director.
Regulation and the Medical Profession–The need for Integrity and Accountability in Physician Leadership and Health Care Policy.
The July 19th, 2011 fax from PHS seen below is in reference to the lab report from USDTL seen above. In it PHS requests the report be “updated”to donor ID number “1310” and to “reflect that the chain of custody was maintained.”
The lab report is a positive test for the alcohol biomarker (Phosphatidyl Ethanol) or PEth, an alcohol biomarker introduced by the Federation of State Physician Health programs and marketed by USDTL and other labs to detect covert alcohol use..
There is no record of where, when or by whom it was collected.
 Both the donor ID # and chain of custody are listed as 461430.
Both the donor ID # and chain of custody are listed as 461430.
The purpose of chain-of-custody is to document the location of a specimen in real time. “Updating” it is not an option. It is prohibited. Updating the “chain of custody to reflect that chain of custody was maintained” is a clear indicator that it was not maintained.
ID #1310 is the unique identifier I was issued by PHS. It is used as a unique identifier, just like a name or social security number, to link me to any sample collected for random drug and alcohol screening. #1310 identifies me as me in the chain-of-custody. On July 1st, 2011 I had a blood test collected at Quest Diagnostics.
The sample was collected at Quest Diagnostics on July 1, 2011 but these documents were not obtained until December 3, 2011 and were included in the “litigation packet” which documents chain-of-custody and is generated on any and all forensic drug testing. It provides proof that the test was done on who it was supposed to have been done and that all required procedure and protocol was followed. It protects the donor form being falsely accused of illicit substance use. In most employee drug-testing programs the litigation-packet is provided on request immediately. It is a transparent process. This is not the case, however, at PHS.
I requested the litigation packet immediately after the positive test was reported on July 19, 2011. PHS first refused, then tried to dissuade me. They finally agreed but warned there would be “unintended consequences. The entire litigation packet can be seen here: Litigation Packet 12:3:2011
The positive sample has no chain-of-custody linked to me, no date, and no indication where it was collected or who collected it. In addition there was no “external” chain of custody for the sample. The custody-and-control form was missing.
With multiple fatal flaws (6/6) rendering it invalid, USDTL should have rejected it by their own written protocol.
USDTL did not reject it. The document below shows that USDTL added my ID # 1310 and added a collection date of July 1, 2011–the day I submitted the sample.
“REVISED REPORT PER CLIENTS REQUEST”
And in doing so the lab that claims “integrity” and “strict chain of custody” readily, and with no apparent compunction” manufactured a chain-of-custody and added a unique identifier by faxed request.
The litigation packet was signed by Joseph Jones on December 3, 2011. There was no record of where the sample was from July 1st to July 8, 2011. No external chain-of-custody or custody-and-control form was evident in the litigation packet.
The V.P. for Laboratory operations for the lab that claims “strict chain of custody” and that “doesn’t skip steps” “when “peoples lives are on the line” verified a positive test as positive with no custody and control form, no external chain of custody and 6/6 fatal flaws. What is so shocking is that this was done without compunction or pause. As a forensic test ordered by a monitoring program Jones knew full well it would result in significant consequences for someone. He knew that someones “life was on the line,” knew it was wrong, and did it anyway.
A person of conscience would never do this. It is unethical decision making that goes against professional and societal norms. A “moral disengagement” that represents a lack of empathy and a callous disregard for others. I would not consider doing something like this for any price and here it appears to be standard operating procedure.
PHS reported the positive test to the Medical Board on July 19, 2011 Positive PEth July 19, 2011-1. It was used as a stepping-stone to request an evaluation at one of three “PHP-approved” facilities (Marworth, Hazelden and Bradford). The Medical Directors of all three facilities can be seen on this list list called “Like-Minded Docs.” The MRO for PHS, Dr.Wayne Gavryck, whose job was to review the chain-of-custody and validate its integrity before reporting it as positive is also on the list. See this simplified schematic of how it works in Massachusetts. It shows how this is a rigged game.
Expecting to be diagnosed with a non-existent problem and admitted for non-needed treatment I requested an evaluation at a non-12 step facility with no conflicts-of-interest. Both PHS and the Medical Board refused this request in one of four violations of the Establishment Clause of the 1st amendment.
I chose Hazelden. The Medical Director was aware that I had just signed a patent license agreement for an epinephrine auto-injector and he had a child with a peanut allergy. We talked about the device and discussed the problems with current management. I think it was because of this added personal interaction that he did not “tailor my diagnosis” as PHS most certainly requested. Seeing me as a person rather than an object, I believe, enabled his conscience to reject it. My discharge diagnosis found no history of alcohol issues but they could not explain the positive test. Unable to rule out that I drank in violation of my PHS contract they recommended I attend AA.
PHS mandated that I attend 3 12-step meetings per week and requested that I obtain names and phone numbers of fellow attendees so they could contact them to verify my attendance. They also mandated that I discontinue my asthma inhalers (as the propellant contains small amounts of ethanol) that had been controlling my asthma and preventing serious attacks for the previous ten years. I was threatened that if I had to use the inhalers or one day late on the increased payments I would be reported to the Board and lose my license.
Sanchez states that my request for the “litigation packet” was processed on December 5, 2011 (two days after Jones signed off on it) and adds the “testing laboratory is willing to support the test results.”
In the interim I filed a complaint with the College of American Pathologists. I also requested the missing external chain of custody documents from Quest.
I never received the chain of custody from Quest. Instead I received a letter from Nina Tobin, Compliance Manager for Quest documenting all the errors but written to sound as if some sort of protocol was maintained. Tobin claimed the specimen was inadvertently logged as a clinical specimen but sent on to USDTL a week later. (See Quest Letter )
The Chief of Toxicology at MGH wrote a letter to the Board documenting all of the misconduct and irregularities stating that it was an “intentional act” perpetrated by PHS. MLLv3finalJacob_Hafter_Esq_copy.
This letter, as well as the opinions of everyone outside of PHS was ignored. So too were any opinions of my two former Associate Directors at PHS. The e-mail below dated October 10th, 2011 is to to Drs.John Knight and J. Wesley Boyd and I am referring to their article Ethical and Managerial Considerations Regarding State Physician Health Programs that was about to be published. We had hoped that it would draw more attention to the problems with PHPs.
I was subsequently reported as “non-compliant” with AA meetings. They could not give any details of where or when. They then misrepresented a declaration of fact (I stated that I had started going to a specific meeting on a specific date) as an admission of guilt by saying I was referring to a different meeting. 10:23:12 PHS Letter to BORM-noncompliance.
My Chief at MGH, his Chief and others held a conference with PHS and attempted to remove me from PHS and replace the monitoring contract with one of their own. They refused. When confronted with the fabricated test they dismissed it and focused on sending me to Kansas to one of the “disruptive physician” Psikhuskas where they are using polygraphs (despite the AMAs stance that it is junk science) and non-validated neuropsychological instruments that detect “character defects” to pathologize the normal.
I refused. Had I gone to Kansas I would have been given a false diagnosis and my career would be over. This is what they do.
Amy Daniels, the investigator for the College of American Pathologists contacted me in December of 2012 to see how things were going since USDTL “amended” the test. Daniels told me that the College of American Pathologists confirmed my allegations and, as an Accrediting Agency for Forensic Toxicology mandated that USDTL correct it. (Labs can lose accreditation if they do not comply with CAP Standards for Forensic Drug Testing). This was done on October 4, 2012.
PHS denied any knowledge of an amended test. I also wrote an e-mail to Joseph Jones requesting the document but he did not reply.
I contacted CAP. On December 11, 2012 Dr. Luis Sanchez wrote a letter stating “Yesterday, December 10 2012, Physician Health Services (PHS) received a revision to a laboratory test result”
“The amended report indicates that the external chain of custody protocol [for that sample] was not followed per standard protocol]”Sanchez dismisses this test as irrelevant, rationalizing neither PHS nor the Board based any actions on the test and they would “continue to disregard” it.
The logic is that it was my behavior that resulted in any consequences. My “non-compliance” in October led to my suspension and the test had nothing to do with it. The sole reason for reporting me to the Board in 2011 was the positive test. There is no other pretext to use. It is misattribution of blame as without the test, now invalidated, there would have been no AA meetings to say I was non-compliant with.
In response to a civil complaint PHS, Quest and USDTL all took the position that the results of the fraudulent testing had absolutely nothing to do with anything.
And in response to the allegations of forensic fraud the labs claimed there was no forensic fraud because this was not a “Forensic” test but a “clinical” test. The argument was that “clinical” tests do not require chain-of-custody and it was his behavior not these tests that resulted in consequences.
As a “clinical” test I knew it was considered Protected Health Information (PHI) under the HIPAA-Privacy Rule. A patient must give written consent for any outside entities to see it. Obtaining lab tests previously required the consent of both the patient and the ordering provider. What PHS and the labs were apparently unaware of was the changes to the HIPAA-Privacy rule giving patients increased rights to access their PHI. The changes removed the ordering provider requirements. A patient has a right to obtain lab test results directly from the labs and has 30 days to do it. CAP agreed. USDTL sent me all of the documents. They can be seen below:
August 6, 2014 to Langan with health materials.
The documents sent by USDTL are notable for two things:
1. The e-mail from me to Joseph Jones dated December 10, 2012. It can be seen on page 22 of the USDTL documents.
2. USDTL document confirming PHS knew the test was amended 67-days before they said they did.
The document shows PHS and Sanchez were aware of the invalidity of the test on October 4, 2012. Instead of correcting things they initiated machinations to throw me under the bus. They officially reported me to the Board for non-compliance on October 19, 2012.
The December 11, 2012 letter signed by Sanchez states “Yesterday, December 10, 2012, PHS received a “revised report” regarding the test. The documents show he knew about it 67-days prior.
Although USDTL complied with the HIPAA-Privacy Rule and CAP, Quest did not. Quest Diagnostics refused to send me copies of their lab reports claiming it was confidential and protected information that required PHS consent. Quest required I sign a consent form with multiple stipulations regarding PHS. I refused and contacted the Department of Justice -Office of Civil Rights. The DOJ-OCR agreed with me and I received the Quest documents
Remember a “clinical” test can only be ordered by a physician in the course of medical treatment. It requires authorization from the patient to obtain a “clinical” specimen and it requires written authorization as to who sees it. Referring physician was Mary Howard.
And below is the fax from PHS to Quest from July 1, 2011 also requested by Mary Howard. The signature on the front is not mine. In addition I gave the blood at 9:30 and was in my clinic at MGH at 12:23 so it couldn’t be. The WC 461430 R are dated July 2, 2011. This is a “clinical” not “forensic” sticker. The “R” indicates a red top tube. The other sticker is USDTL and indicates it was logged in on July 8, 2011.
What does it all mean? Blood left in a red top tube ferments. This is basic chemistry. The PEth test needs to be refrigerated and shipped overnight to prevent this. In addition it needs to be collected with a non-alcohol wipe in a tube that has an anti-coagulant or preservative so that it does not ferment. It requires strict procedure and protocol.
When I gave my blood on July 1st, 2011 it was as a “forensic” test per my contractual agreement with PHS.
On July 2, 2011 it was changed to “clinical.” Why? because “forensic” protocol would have invalidated it.
The only conceivable reason for doing this was to bypass chain-of-custody procedures. My unique identifier #1310 was removed and the clinical specimen number was used for chain-of-custody. The R in 461430R indicates a red top tube.
By holding on to it for one week the blood fermented. As it was July with an average temperature close to 90 they overshot their mark a bit. My level of 365 is consistent with heavy alcohol use–end stage half-gallon a day type drinking.
Quest then forwarded it to USDTL with specific instructions to process it as a “clinical” sample. USDTL complied and processed it as a clinical specimen which was reported it to PHS on July 14, 2011.
PHS then asked USDTL to add my forensic ID # 1310 and add a collection date of July 1, 2011 so it would appear “forensic” protocol was followed. The reason Jones signed the “litigation packet” on December 3, 2011 was because that was when the “litigation packet” was manufactured. A “clinical” sample does not produce one.
USDTL willingly complied with this request.
PHS then reported this as a “forensic” test to the Medical Board on July 19, 2011 and requested a reevaluation.
The distinction between “forensic” and “clinical” drug and alcohol testing is black and white. PHS is a monitoring program not a treatment provider. The fact that a monitoring agency with an MRO asked the lab to process and report it as a clinical sample and then used it forensically is an extreme outlier in terms of forensic fraud. The fact that they collected it forensically, removed the forensic components and let it sit in a warehouse for a week is abhorrent. The fact they then specifically requested it be processed as a clinical sample deepens the malice. The fact that they then reported it to the Board as a forensic sample and maintained it was forensic up until just recently makes it egregious. But the fact that the test was changed from “positive” to “invalid” on October 4th, 2012 and they then reported me to the Board on October 8th, 2012 for “noncompliance,” suppressed it and tried to send me to Kansas where I would be given a non-existent diagnosis to delegitimize me for damage control makes it wantonly egregious. This is political abuse of psychiatry.
Accountability requires both the provision of information and justification of what was done.
For doctors it is very difficult to obtain the information. As seen here, they put up a gauntlet to prevent the provision of what is immediate in all other drug testing programs. I now have all of the information. What it shows is clear. This was intentional. It was no accident. They knew what they were doing, knew it was wrong but did it anyway.
Accountability also requires that those who commit misconduct suffer consequences. The PHPs have also put up barriers to this. With no regulation or oversight they have no apparent accountability.
My understanding is that it works this way. The Medical Board, Medical Society and Departments of Public Health have no oversight. The MMS has an ethics committee but all they can do is “educate” the person if they feel there was a violation. The DPH won’t even look at it and the Board is complicit.
My understanding is that they have convinced law enforcement that this is a “parochial” issue that is best kept within the medical community. They have also created the impression that they are “friends” of law enforcement. I have heard from many doctors that they have tried to report misconduct, civil rights violations and crimes to the police, AGO, and other law enforcement agencies only to be turned back over to the PHP. By saying the physician is “impaired” it delegitimizes and invalidates the truth. “He’s just a sick doctor, we’ll take care of him.” That physician then suffers consequences effectively silencing the rest.
PHS uses the Board to enforce punitive measures and temporize. The Board puts blind faith in PHS. Blind faith that defies common sense ( mandating phone numbers at anonymous meetings) and disregards the law (Establishment Clause violations that are clear and well established). The Board also temporizes to cause damage.
In my case they required a psychiatric behavioral evaluation. I was given the choice of Kansas and a few other Like-minded assessment centers.
After petitioning for multiple qualified psychiatrists that were summarily rejected months later for no reason one of the Board Attorneys suggested Dr. Patricia Recupero, M.D., J.D. who is Board Certified in Forensic Psychiatry and Addiction Psychiatry. The Board had used her in the past but not recently. Seeing that she had been used by the Board for fit-for-duty evaluations in the past the Board accepted my petition.
Dr. Recupero wrote an 87-page report. She concluded I was safe to practice medicine without supervision, that I had never had an alcohol use, abuse or dependence problem, and that PHS request for phone numbers was inappropriate. She also documented PHS misconduct throughout my contract and concluded it was PHS actions, not mine, that led to my suspension. What she describes is consistent with criminal harassment. She documents the falsification of neuropsychological tests and confirms the forensic fraud. What did the Board do? Ignored their very own recommended and approved evaluator.
One measure of integrity is truthfulness to words and deeds. These people claim professionalism, ethics and integrity. The documents show otherwise. The careers and lives of doctors are in these peoples hands.
Similar fraud is occurring across the country. This is an example of the institutional injustice that is killing physicians. Finding themselves entrapped with no way out, helpless and hopeless they are feeling themselves bereft of any shade of justice and killing themselves. These are nothing more than bullies and accountability is essential. The “disruptive physician” moral panic has harmed the Medical Profession.
Dr. Clive Body in his book Corporate Psychopaths writes that “Unethical leaders create unethical followers, which in turn create unethical companies and society suffers as a result.” And according to Dr. Robert Hare in Without Conscience “If we can’t spot them, we are doomed to be their victims, both as individuals and as a society. ”
Wes Boyd notes that valid complaints from physicians are often dismissed as “bellyaching” by the PHPs. Complacent that these are just good guys helping doctors and protecting the public the complaints are dismissed, tabled, deflected or otherwise ignored. Bellyaching?? Is this bellyaching.
It is my opinion that what you see here is indefensible Procedurally, Ethically, and Legally.
Procedurally it goes beyond negligence and represents fraud. It violates every procedural guideline, regulation and standard of care including their very own.
Ethically it violates everything from the Hippocratic Oath to AMA Medical Ethics to the MRO Code of Conduct.
And where was PHS MRO Wayne Gavryck? By my count he violated at least 4 of the 6 Codes of Ethical Conduct.
What was done here violates the most fundamental ethical principles of Medicine -Autonomy, Beneficence, Nonmaleficence and justice.
Intentionally falsifying a laboratory or diagnostic test to refer for an evaluation or support a diagnosis or give unwarranted “treatment” is unconscionable. Abuse under the utility of medical coloration is especially egregious.
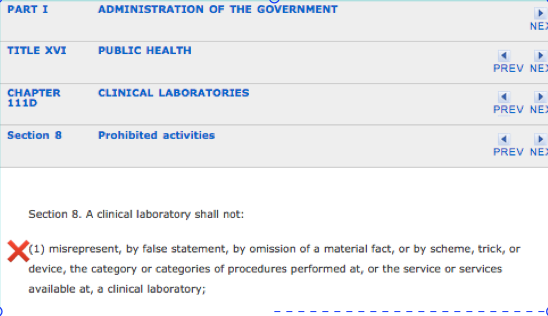
The information provided herein should negate any “peer-review” protection or immunity afforded PHS as it is undeniably and egregiously in “bad faith.” Moreover, the ordering a “clinical” test is outside PHS scope, practice, and function of PHS. According to M.G.L. c. 111, § 203 (c):
An individual or institution, including a licensed or public hospital, physician credentialing verification service operated by a society or organization of medical professionals for the purpose of providing credentialing information to health care entities, or licensed nursing home reporting, providing information, opinion, counsel or services to a medical peer review committee, or participation in the procedures required by this section, shall not be liable in a suit for damages by reason of having furnished such information, opinion, counsel or services or by reason of such participation, provided, that such individual or institution acted in good faith and with a reasonable belief that said actions were warranted in connection with or in furtherance of the function of said committee or the procedures required by this section.
Dr. Luis Sanchez and Dr. Wayne Gavryck need to be held to the same professional standards as the rest of us.
If you can support either of them procedurally, ethically, or legally, any one of them, then I will turn in my medical license with a bow on it. If they did not commit negligent fraud by standards of care and procedural guidelines, egregious moral disengagement in violation of ALL ethical codes for the medical profession and society and break the law then disprove me. Just one will do.
But you can’t do this then I ask that you speak up and take a stand. Either defend them or help me hold them accountable. If a crime is committed it needs to be addressed. Ignoring encourages more of the same.
And if this cannot be supported procedurally, ethically or legally then I want to know what is going to be done about it?
How low does the moral compass have to go before someone takes action?
Doctors are dying across the country because of people just like this. They have set up a scaffold that removes the usual checks and balances and removed accountability. It is this institutional justice that is driving many doctors to suicide.
So the evidence is above. Either defend them or help me draw unwanted attention to this culture of bullying and abuse. So I am asking you to contemplate if what you see here is ethically, procedurally or legally sound. If you can show just one of these then I stand corrected. But if you cannot justify this on any level then I want you to help me expose this criminal enterprise. Either defend it or fight it. Silence and obfuscation are not acceptable.




































 The Plan to… Require Doctors to Drug-Test all Patients
The Plan to… Require Doctors to Drug-Test all Patients








 The Elephant in the room is the state Physician Health Programs organized under the FSPHP. Nothing has changed–they have only grown more powerful and opaque and removed themselves from accountability and culpability. Moreover, they are expanding to other fields. Just ask the airline pilots. They eventually want to expand to
The Elephant in the room is the state Physician Health Programs organized under the FSPHP. Nothing has changed–they have only grown more powerful and opaque and removed themselves from accountability and culpability. Moreover, they are expanding to other fields. Just ask the airline pilots. They eventually want to expand to