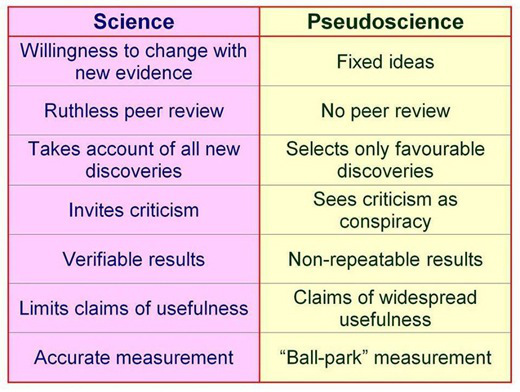
It is only a few public policy steps and minor changes in state regulatory statutes before what is described in the ASAM White Paper on Drug Testing comes to fruition. Before we know it the Drug and Alcohol Testing Industries “New Paradigm” as described here by Robert Dupont will be ushered in. From the ASAM white Paper:
“THIS WHITE PAPER ENCOURAGES WIDER AND “SMARTER” USE OF DRUG TESTING WITHIN THE PRACTICE OF MEDICINE AND, BEYOND THAT,BROADLY WITHIN AMERICAN SOCIETY. SMARTER DRUG TESTING MEANS INCREASED USE OF RANDOM TESTING* RATHER THAN THE MORE COMMON SCHEDULED TESTING,* AND IT MEANS TESTING NOT ONLY URINE BUT ALSO OTHER MATRICES SUCH AS BLOOD, ORAL FLUID (SALIVA), HAIR, NAILS, SWEAT AND BREATH WHEN THOSE MATRICES MATCH THE INTENDED ASSESSMENT PROCESS. IN ADDITION, SMARTER TESTING MEANS TESTING BASED UPON CLINICAL INDICATION FOR A BROAD AND ROTATING PANEL OF DRUGS RATHER THAN ONLY TESTING FOR THE TRADITIONAL FIVE-DRUG PANEL.”Backed by the multi-billion dollar drug and alcohol testing, assessment and treatment industry the public policy positions of the American Society of Addiction Medicine (ASAM) have invariably passed. There has been little if any meaningful opposition.
To prevent this future drug testing dystopia, that includes testing schoolchildren, we need to take a step back and analyze the reliability and credibility of the “evidence-base” behind these multiple non-FDA approved (Introduced as Laboratory Developed Tests (LDTs) to bypass FDA approval) “forensic” drug and alcohol tests and testing devices (The alcohol biomarkers EtG, EtS, PEth; SCRAM (Subcutaneous Remote Alcohol Monitoring Bracelet);CDPB (Cellular Digital Photo Breathalyzer); and Hair Testing- Psychemedics, etc.) the ASAM proposes be used on the population at large. These tests include nail, hair, saliva, breath, blood and urine and they plan on utilizing the Medical Profession as a urine collection agency by calling this testing a “medical evaluation” rather than “monitoring” for drug and alcohol use. This change in semantics enables them to bypass the usual forensic drug testing protocol (that includes strict chain-of-custody collection and MRO review) designed to minimize false-positives because the results of erroneous test can be grave and far reaching. According to the ASAM white paper the “clinical” collection of specimens as is good enough as the results of a positive test will result in “treatment” rather than “punishment.”
Amazingly, there has been no Academic review of these tests, let alone a Cochrane type critical analysis. It is essentially untapped territory. In addition there has been no Institute of Medicine type Conflict of Interest Analysis.
And that is why I am asking for help from statisticians, biostatisticians and epidemiologists. The task would entail a review of the literature prior to the introduction of these tests for evidence base of forensic applicability (there essentially is none) and a review of the literature peri-and post marketing of these devices to assess the reliability and credibility of the underlying methodology and ascertain the evidence-base. The goal would be publication in both academic journals and presentation to the general public through media publication with the assistance of investigative journalists and other writers. The goal is to get the truth out about these tests and allow both the medial profession and public at large to awaken to the menace this represents. I can’t pay you but you would be combating injustice, corruption and dishonesty. You would be doing your part in helping the Medical Profession, honest and decent doctors, our country and perhaps our future.